InMed Pharmaceuticals Reports Second Quarter Fiscal 2021 Financial Results

VANCOUVER, BC – InMed Pharmaceuticals Inc. (“InMed” or the “Company”) (NASDAQ:INM; TSX:IN), a clinical-stage pharmaceutical company developing medications targeting diseases with high unmet medical need and leading the way in the clinical development of cannabinol (“CBN”), today reported financial results for the second quarter of fiscal year 2021 (“2Q21”) which ended December 31, 2020.
Conference Call & Webcast1:
Thursday, February 11, 2021 at 8:30 AM Pacific Time, 11:30 AM Eastern Time
Local – Toronto (+1) 416-764-8609
Local – Vancouver (+1) 778-383-7417
Toll Free – North America (+1) 888-390-0605
Conference ID: 19917866
Webcast: https://produceredition.webcasts.com/starthere.jsp?ei=1425283&tp_key=9f171376cb
(1Webcast replay available for 90 days)
Dial-In Replays2:
Toronto: (+1) 416-764-8677
North America (Toll Free): (+1) 888-390-0541
Playback Passcode: 917866 #
(2Dial-in replay available till February 18, 2021)
The Company’s 10-Q including financial statements and related MD&A for the second quarter ending December 31, 2020 are available at www.inmedpharma.com, www.sedar.com and at www.sec.gov.
“The second quarter of fiscal 2021 saw InMed achieve several key, value-adding and risk mitigating milestones across both its therapeutic programs and IntegraSynTM manufacturing approach,” said InMed President and Chief Executive Officer, Eric A. Adams. “We now have a strong body of safety and tolerability data for INM-755, which is the basis for our plans to commence safety and efficacy trials in Epidermolysis Bullosa patients this calendar year. We announced business development activities in both our ocular therapeutic and our innovative, biosynthesis-based IntegraSynTM manufacturing program this last quarter. These announcements are a reflection of our increased emphasis on business development activities as an avenue to advance our R&D efforts.”
Research & Development Update:
INM-755 for the Treatment of Epidermolysis Bullosa (“EB”):
- On November 25, 2020, InMed announced top-line results from its 755-101-HV Phase 1 clinical trial (“Study 101”). Study 101 was a randomized, vehicle-controlled, double-blind, Phase 1 trial, that examined the safety and tolerability of two strengths of INM-755 cream on intact skin in 22 healthy adult volunteers over a 14-day treatment period. The Study 101 results indicate that INM-755 was safe and well-tolerated on intact skin, caused no systemic or serious adverse effects. In addition, there were no subject withdrawals due to adverse events. Drug concentrations in the blood were very low, as expected.
- In addition, on January 8, 2021, we announced top-line results from our second Phase 1 clinical trial, 755-102-HV (“Study 102”). Study 102 was a randomized, double-blind, vehicle controlled, single-center study, in 8 healthy adult volunteers to test the tolerability of 14 days of application of the INM-755 cream on epidermal wounds under treatment procedures designed to simulate wound care for Epidermolysis Bullosa (“EB”) patients with open wounds. Results of Study 102 indicate that INM-755 cream was safe and well-tolerated on induced open epidermal wounds, caused no systemic or serious adverse effects. In addition, there were no subject withdrawals due to adverse events.
- These data from Study 101 and Study 102 support moving forward into clinical trials in patients with EB. Regulatory applications for a Phase 2 safety and efficacy study in patients with EB are in preparation with filings in several countries expected in the first half of calendar 2021, and with patient enrollment anticipated to begin in the second half of the calendar year.
INM-088 for the Treatment of Glaucoma:
- On December 3, 2020, we announced securing an exclusive, worldwide license from EyeCRO LLC for its Microemulsion Drug Ocular Penetration System (“MiDROPS®”) eyedrop delivery technology targeting effective, topical administration of all cannabinoids, cannabinoid variants and analogs to the eye. MiDROPS® is a proprietary platform technology designed to effectively deliver lipophilic molecules to both the anterior and posterior segments of the eye by means of a stable and comfortable eyedrop formulation. InMed has completed a preliminary investigation demonstrating simplicity and effectiveness of MiDROPS® to deliver sustained levels of CBN to the eye for the prospective treatment of ocular diseases, including glaucoma. With this licensing agreement in place, InMed anticipates initiation of IND-enabling toxicology studies with INM-088 formulation, MiDROPS® incorporating CBN, in 2021.
IntegraSynTM:
- On November 18, 2020, InMed announced a broad, reciprocal research collaboration with BayMedica Inc. to explore synergies between technologies owned by the two companies. Under the agreement, BayMedica will research the potential benefits of one or more of the high-efficiency enzyme gene sequences from InMed’s proprietary IntegraSynTM platform for production of cannabinoids. In exchange, InMed is being granted access to selected cannabinoid analog compounds from BayMedica’s extensive library to explore their therapeutic potential in selected disease models in the field of neuroprotection.
Financing Activity and Results of Operations (expressed in US Dollars):
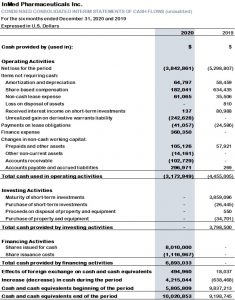
- On November 16, 2020, the Company successfully closed a public offering of an aggregate of 1,780,000 common shares, together with accompanying warrants, at a public offering price of $4.50 per share and accompanying warrant for total gross proceeds of approximately $8M. Commensurate with the pricing of the offering, the Company’s shares began trading on the Nasdaq Capital Market under the trading symbol “INM”.
- On February 5, 2021, the Company announced a private placement with certain institutional investors to raise aggregate gross proceeds of approximately $4.5 million at a price of $4.25 per unit consisting of one common share and 0.66 of a warrant to purchase one common share. The offering, which is subject to TSX and Nasdaq approval, is anticipated to close on or about February 12, 2021. InMed intends to use the proceeds from the offering, together with its existing cash resources, for general corporate purposes, funding preclinical and clinical development programs, advancing IntegraSynTM, and for working capital purposes.
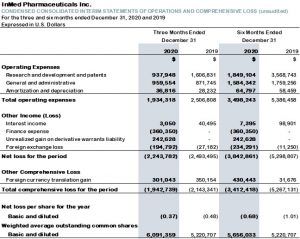
- For the three and six months ended December 31, 2020, the Company recorded a net loss of $2.2 million and $3.8 million, or $0.37 and $0.68 per share, compared with a net loss of $2.5 million and $5.3 million, or $0.48 and $1.01 per share, for the three and six months ended December 31, 2019.
- Research and development expenses were $0.9 million for 2Q21, compared with $1.6 million for the three months ended December 31, 2019. For the six months ended December 31, 2020, research and development expenses totaled $1.8 million, which compares with $3.6 million for the comparable period in fiscal 2020. The reduction in research and development expenses in both the three months and six months ended December 31, 2020 was primarily due to decreased spending on the integrated cannabinoid manufacturing program and the INM-755 program, which completed its Phase 1 studies at the end of the period. In addition, share-based payments were lower and purchases of the active pharmaceutical ingredient used in INM-755 clinical trials decreased.
- The Company incurred general and administrative expenses of $1.0 million for 2Q21, compared with $0.9 million for the three months ended December 31, 2019. For the six months ended December 31, 2020, general and administrative expenses totaled $1.6 million, which compares with $1.8 million for the comparable period in fiscal 2020. The decrease in general and administrative expenses for the six months ended December 31, 2020 results from a combination of changes including lower share-based payments and lower legal costs associated with negotiating research and development contracts and other matters in the current period and certain current year legal costs being expensed as finance expense or capitalized to equity, offset by substantially higher insurance fees. In addition, a decrease in personnel resulted in lower salaries and benefits.
- The Company also incurred non-cash, share-based payments, in connection with the grant of stock options, of $0.1 million for 2Q21, compared with $0.3 million for the three months ended December 31, 2019. For the six months ended December 31, 2020, non-cash, share-based payments totaled $0.1 million, which compares with $0.6 million for the comparable period in fiscal 2020. Share-based payments amounts are included within research and development expenses and general and administrative expenses.
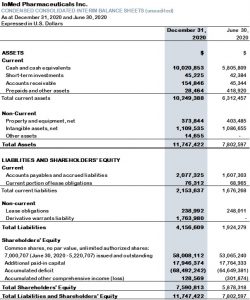
- At December 31, 2020, the Company’s cash, cash equivalents and short-term investments were $10.1 million, which compares to $4.5 million at September 30, 2020 and $5.8 million at June 30, 2020. The increase in cash, cash equivalents and short-term investments during the six months to December 31, 2020, was primarily the result of the November 16, 2020 public offering partially offset by cash outflows from operating activities.
- At December 31, 2020, the Company’s total issued and outstanding shares were 7,000,707. Including outstanding stock options and warrants, as at December 31, 2020, the Company had 9,676,774 shares on a fully diluted basis. During the three and six months ending December 31, 2020, the weighted average number of common shares was 6,091,359 and 5,656,033, which is used for the calculation of loss per share for the respective interim periods.
Table 1: Condensed consolidated interim balance sheets (unaudited):
Table 2: Condensed consolidated interim statements of operations and comprehensive loss (unaudited):
Table 3: Condensed consolidated interim statements of cash flows (unaudited):
About InMed: InMed Pharmaceuticals is a clinical-stage pharmaceutical company developing a pipeline of cannabinoid-based medications, initially focused on the therapeutic benefits of cannabinol (CBN) in diseases with high unmet medical need. The Company is dedicated to delivering new therapeutic alternatives to patients that may benefit from cannabinoid-based medicines. For more information, visit www.inmedpharma.com.
Investor Contact:
InMed Pharmaceuticals Inc.
Brendan Payne, Director – Investor Relations
T: +1.604.669.7207
Edison Advisors for InMed Pharmaceuticals
Joe Green/Laine Yonker
T: +1.646.653.7030/+1.646.760.0321
E: jgreen@edisongroup.com / lyonker@edisongroup.com
Cautionary Note Regarding Forward-Looking Information:
This news release contains “forward-looking information” and “forward-looking statements” (collectively, “forward-looking information”) within the meaning of applicable securities laws. Forward-looking information is based on management’s current expectations and beliefs and is subject to a number of risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Forward-looking information in this news release includes statements about: leading the way in the clinical development of cannabinol (“CBN”); developing a pipeline of cannabinoid-based medications in diseases with high unmet medical need; delivering new therapeutic alternatives to patients that may benefit from cannabinoid-based medicines; commencing safety and efficacy trials in Epidermolysis Bullosa patients in calendar 2021; increasing business development activities to advance our research and development efforts; filing of regulatory applications for a Phase 2 safety and efficacy study in patients with EB in several countries in the first half of calendar 2021, and commencing patient enrollment the second half of the calendar year; being able to deliver sustained levels of CBN to the eye for the prospective treatment of ocular diseases; initiating IND-enabling toxicology studies with INM-088 formulation, MiDROPS® incorporating CBN, in 2021; exploring the therapeutic potential of BayMedica Inc.’s cannabinoid analog compounds in selected disease models in the field of neuroprotection; and the closing of the private placement, announced on February 5, 2021, on or about February 12, 2021 or at all.
With respect to the forward-looking information contained in this news release, InMed has made numerous assumptions regarding, among other things: the closing of the private placement on a timely basis on the terms described, or at all; continued and timely positive preclinical and clinical efficacy data; the speed of regulatory approvals; the ability to contract with suitable partners; demand for InMed’s products; and continued economic and market stability. While InMed considers these assumptions to be reasonable, these assumptions are inherently subject to significant business, economic, competitive, market and social uncertainties and contingencies.
Additionally, there are known and unknown risk factors which could cause InMed’s actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking information contained herein. Known risk factors include, among others: regulatory filings may not be filed or approved on a timely basis, or at all; our securities issuable in connection with the private placement may not be accepted for registration by the Securities and Exchange Commission on a timely basis, or at all; the outbreak and impact of COVID-19 may worsen; preclinical and clinical testing may not produce the desired results on a timely basis, or at all; cannabis licensing/importing issues may delay our projected development timelines; suitable partners may not be located; economic or market conditions may worsen; our existing cash runway may not allow us to complete our forthcoming significant milestones; the development of a proprietary biosynthesis manufacturing technology for the production of pharmaceutical-grade cannabinoids as well as a pipeline of medications targeting diseases with high unmet medical needs may not be as successful as desired, if at all. A more complete discussion of the risks and uncertainties facing InMed is disclosed in InMed’s filings with the Security and Exchange Commission and the most recent Annual Information Form and other continuous disclosure filed with Canadian securities regulatory authorities on SEDAR at www.sedar.com.
All forward-looking information herein is qualified in its entirety by this cautionary statement, and InMed disclaims any obligation to revise or update any such forward-looking information or to publicly announce the result of any revisions to any of the forward-looking information contained herein to reflect future results, events or developments, except as required by law.
NEITHER THE TORONTO STOCK EXCHANGE NOR ITS REGULATIONS SERVICES PROVIDER HAVE REVIEWED OR ACCEPT RESPONSIBILITY FOR THE ADEQUACY OR ACCURACY OF THIS RELEASE.